MiraChlor™ delivers 100% natural, pharmaceutical-grade Hypochlorous Acid (HOCl) — the same molecule produced by your patient's neutrophils — for biofilm-targeted oral care without alcohol, chlorhexidine, or chemical surfactants.
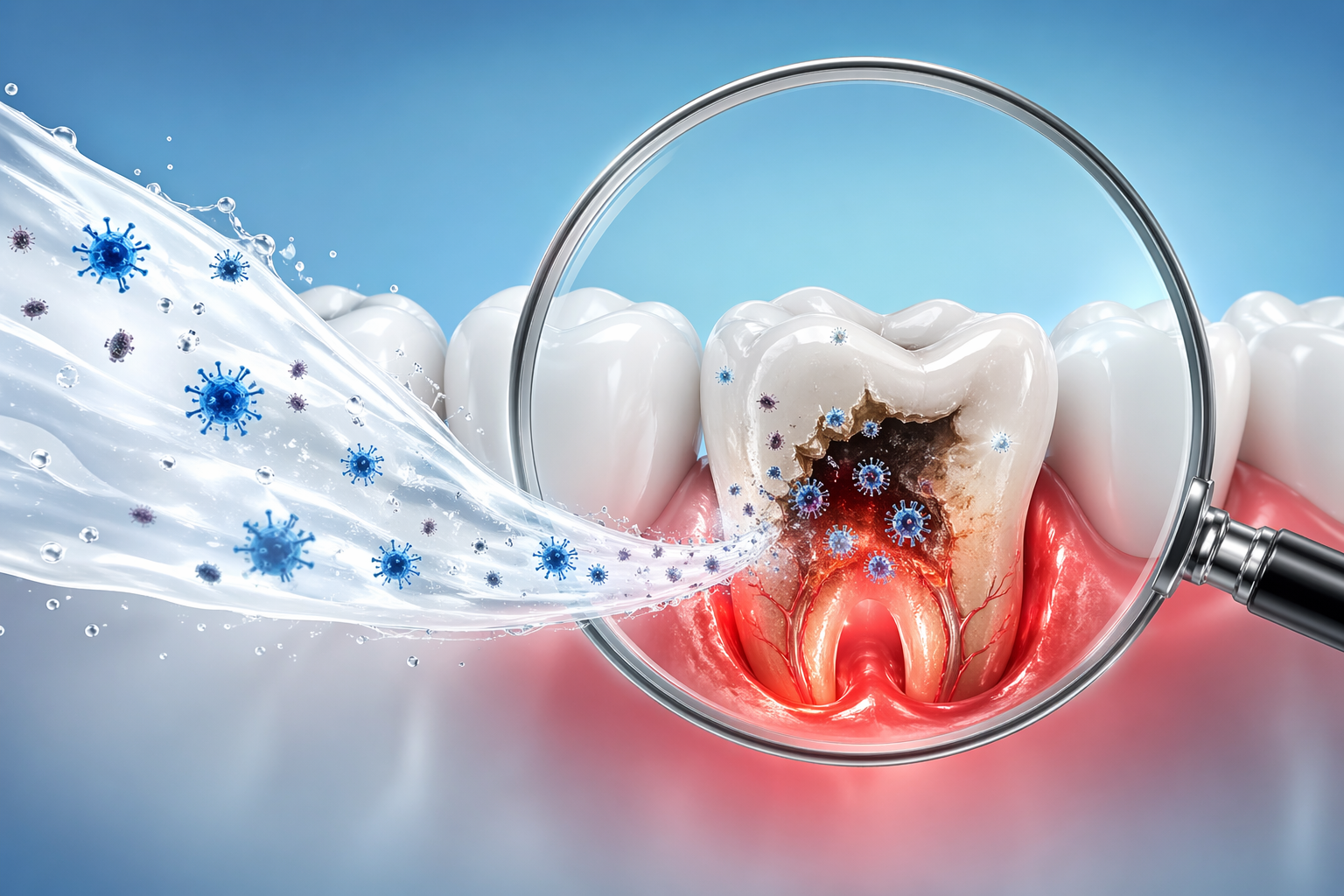
Hypochlorous Acid is produced endogenously by neutrophils as a first-line antimicrobial response. MiraChlor™ delivers it pharmaceutical-grade and pH-stabilized to the oral cavity — disrupting mature biofilm without binding to soft tissue or staining enamel like cationic antiseptics.
What the chemistry does, expressed clinically — selectivity, penetration, pH window, and resistance profile.
HOCl penetrates microbial cell walls and oxidizes essential proteins. Eukaryotic cells, with their thicker membranes and antioxidant systems, remain intact — a selectivity unique among antimicrobials.
Mature plaque biofilms hide pathogens behind an extracellular polysaccharide scaffold. HOCl penetrates and dissolves the matrix where chlorhexidine and CPC plateau on contact.
Buffered at pH 4.5–5.5 — the range that maintains HOCl in its protonated, active form without contributing to enamel demineralization. No acidic erosion. No alkaline tissue burn.
Oxidative mechanisms target multiple cellular components simultaneously, leaving no single pathway for organisms to mutate around — unlike sub-MIC chlorhexidine exposure, which has documented resistance pressure.
MiraChlor™ is supplied in two formulations — a prescription-strength solution cleared by the FDA for in-office use, and an oral rinse calibrated for safe daily and post-operative care.

For in-operatory use during surgical and clinical procedures. Higher-concentration HOCl delivered chair-side by the dental team.

Patient-strength formulation for take-home use. Calibrated for daily and post-operative care without alcohol sting, taste alteration, or extrinsic staining.
Independent of marketing claims — what does the chemistry tell you about long-term tolerability and efficacy?
| Property | MiraChlor™ HOCl | Alcohol-based rinse | Chlorhexidine 0.12% |
|---|---|---|---|
| Active mechanism | Oxidative (mimics neutrophil response) | Membrane disruption (14–26% ethanol) | Cationic surfactant binding |
| Burning sensation | None | Significant | Mild |
| Tooth / restoration staining | None reported | Mild | Extrinsic staining |
| Taste alteration | None | Brief | Documented dysgeusia |
| pH | 4.5–5.5 (enamel-safe) | 4.0–6.0 | 5.5–6.0 |
| Daily long-term use | Indicated | Not recommended | 2-week guideline cap |
| Soft-tissue toxicity | None reported | Mucosal dryness | Cytotoxic to fibroblasts (in vitro) |
| Origin | 100% natural HOCl | Synthetic ethanol formulation | Synthetic biguanide |
Properties drawn from peer-reviewed literature on each active class. Full DOI references available in our Practitioner Brief — request below.
Manufactured in an FDA-registered facility under cGMP protocols. Independent stability and efficacy data on file — full citations in the Practitioner Brief.
Manufactured in a registered facility under cGMP protocols.
End-to-end domestic manufacturing chain.
Peer-reviewed sources backing every clinical claim.
20-page protocol guide and DOI list — request access.
We swapped chlorhexidine for MiraChlor in our peri-implant maintenance protocol. The tissue response in the first 30 days has been notably better — and patients aren't asking why their crowns turned brown.
First rinse I've found that hygienists, periodontists, and post-op patients all tolerate. No sting on raw tissue. No taste complaints. We carry it on every operatory tray now.
Pre-procedural rinse with MiraChlor is now standard for any aerosol-generating procedure in my surgery. The chemistry — endogenous HOCl, pH-buffered — is exactly what I'd want a patient swallowing residue of.